The GLP-1 Race in 2026: Lilly vs. Novo Nordisk and the Next Wave of Weight Loss Drugs
The Two-Company Market: Q1 2026 Head to Head
Key Numbers
Total Revenue: $19.8B (+56% YoY)
Adjusted EPS: $8.55 (+156%)
Mounjaro: $8.66B (+100%+ YoY)
Zepbound: $4.16B (+80% YoY)
US Market Share: 60.1%
2026 Guide: $82-85B (raised $2B)
Total Revenue: $10.8B (beat estimates)
Adjusted EPS: $1.03 (beat)
Wegovy: DKK 18.2B (+12% YoY)
Wegovy Pill: $354M (doubled estimates)
US Market Share: 39.4%
2026 Guide: Adjusted sales decline 4-12% at CER
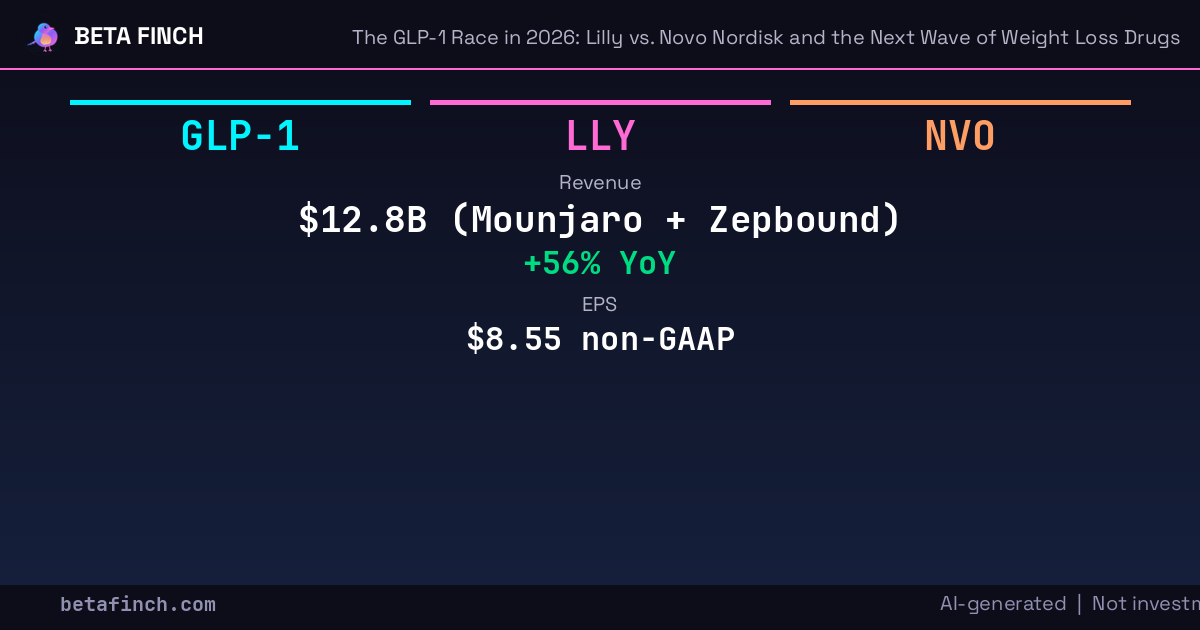
The GLP-1 market in 2026 is a two-company duopoly with a widening gap. Eli Lilly (LLY) reported Q1 2026 revenue of $19.8 billion, up 56% year over year, with adjusted EPS of $8.55, up 156%. Novo Nordisk (NVO) posted revenue of $10.8 billion, beating estimates but guiding for a 4% to 12% decline in adjusted sales at constant exchange rates for the full year. Lilly raised full-year guidance by $2 billion. Novo narrowed its range but kept the word decline in the outlook.
The US market share split tells the story most directly: Lilly holds 60.1% of the combined obesity and diabetes GLP-1 market. Novo holds 39.4%. A year ago, the gap was narrower. What changed was tirzepatide.
Eli Lilly: Three Products, One Molecule, Total Dominance
Lilly's GLP-1 franchise is built on tirzepatide, a dual GLP-1/GIP receptor agonist, sold as Mounjaro for type 2 diabetes and Zepbound for obesity. Combined revenue from those two products reached $12.8 billion in Q1 2026, representing $6.7 billion in year-over-year growth. Mounjaro alone generated $8.66 billion, with international expansion driving the top line as the drug achieved 53% market share outside the US.
The third product is Foundayo (orforglipron), the first and only oral GLP-1 receptor agonist approved in the US. The FDA approved it on April 1, 2026, and Lilly launched on April 6. By the end of the first few weeks, over 20,000 patients had started treatment, with 80% new to the GLP-1 class. That last detail matters: Foundayo is not cannibalizing injectable patients. It is expanding the market. Pricing was set at $149 per month for self-pay patients, $25 with commercial insurance, and $50 for Medicare beneficiaries starting July 1.
Novo Nordisk: The Wegovy Pill Surprise and the CagriSema Setback
Novo Nordisk's Q1 2026 results contained one clear win and one significant loss. The win: the Wegovy pill (oral semaglutide 25 mg), launched January 5, 2026, generated $354 million in its first full quarter, doubling analyst estimates. The drug attracted 1.3 million prescriptions in Q1 and over 2 million since launch, making it the strongest-ever volume launch for a GLP-1 in the US.
The loss: CagriSema, Novo's next-generation combination of cagrilintide and semaglutide, failed its primary endpoint in the REDEFINE 4 head-to-head trial against tirzepatide. Over 84 weeks in 809 patients, CagriSema delivered 23% mean weight loss versus 25.5% for tirzepatide. The trial was designed to prove non-inferiority. It did not. A US regulatory decision is still expected by end of 2026, but the competitive narrative has shifted. Novo's flagship pipeline candidate is demonstrably less effective than Lilly's already-marketed drug.
Ozempic, the diabetes franchise anchor, saw sales decline 8% year over year, though the result beat lowered expectations. The overall obesity care segment grew 22% at constant exchange rates, carried almost entirely by the Wegovy pill launch.
The Efficacy Ladder: How Every GLP-1 Compares
Cross-trial comparisons are imperfect because patient populations, durations, and baseline weights differ. But the directional picture is clear, and two true head-to-head trials anchor the data. In REDEFINE 4, tirzepatide delivered 25.5% weight loss versus CagriSema at 23% over 84 weeks. Lilly's triple agonist retatrutide posted 28.7% weight loss in TRIUMPH-4 Phase 3, the highest figure from any GLP-1-class drug to date.
Among injectables, Roche's CT-388 (enicepatide) showed 22.5% weight loss in Phase 2 at 48 weeks with no plateau reached, and Amgen's MariTide reached 20% at 52 weeks in Phase 2. Among orals, Structure Therapeutics' aleniglipron led with 16.3% weight loss at 44 weeks, followed by Novo's Wegovy pill at 16.6% at 64 weeks, and Lilly's Foundayo at 12.4% at 72 weeks. Viking's oral VK2735 showed 12.2% at just 13 weeks in Phase 2, making a direct comparison premature.
The Challengers: Five Companies Racing to Phase 3
Behind the Lilly-Novo duopoly, five companies are building GLP-1 programs with distinct competitive angles.
Amgen's MariTide (maridebart cafraglutide) is an antibody-peptide conjugate targeting GLP-1 and GIP receptors. Its differentiator is dosing frequency: monthly or every eight weeks, versus weekly for existing injectables. Amgen has enrolled over 3,500 patients in its MARITIME Phase 3 program, with readouts expected in early 2027. In Q1 2026, Amgen announced a SWITCH study enrolling 300 patients transitioning from weekly semaglutide or tirzepatide to MariTide on less frequent schedules, positioning the drug as a potential maintenance or step-down therapy. FDA approval is realistically a 2029 to 2030 event.
Roche entered through its $2.7 billion acquisition of Carmot Therapeutics. CT-388 (enicepatide), a dual GLP-1/GIP injectable, delivered 22.5% weight loss in Phase 2 with 54% of patients on the highest dose achieving a BMI under 30. Roche initiated two Phase 3 trials (ENITH-1 and ENITH-2) in Q1 2026 and plans a combination study with petrelintide, an amylin analog, in mid-2026. Roche is also developing CT-996, an oral GLP-1 in Phase 2, with a readout expected before year-end.
Viking Therapeutics has both injectable and oral versions of VK2735, a dual GLP-1/GIP agonist. The injectable Phase 3 program (VANQUISH) has enrolled over 5,500 patients across two trials, with enrollment completed ahead of schedule. The oral version showed 12.2% weight loss at 13 weeks in Phase 2, and Phase 3 is expected to begin in Q4 2026. Viking is a clinical-stage company with no product revenue.
Structure Therapeutics' aleniglipron, an oral small-molecule GLP-1 agonist, posted the highest efficacy among oral candidates: 16.3% weight loss at 44 weeks in Phase 2 with only 2% discontinuation due to side effects. Registrational Phase 3 is expected to start in Q3 2026. Roche paid Structure $100 million for a non-exclusive patent license related to its oral GLP-1 technology. Multiple analysts have flagged Structure as a potential acquisition target.
Pfizer exited the race. The company discontinued danuglipron, its oral GLP-1 candidate, in April 2025 after a case of potential drug-induced liver injury. Pfizer retains earlier-stage obesity programs but is no longer a near-term competitor.
Lilly's Pipeline: Retatrutide and the Triple Agonist Thesis
The most closely watched pipeline asset in the GLP-1 space is Lilly's retatrutide, a triple agonist targeting GLP-1, GIP, and glucagon receptors simultaneously. TRIUMPH-4 Phase 3 data showed 28.7% mean weight loss (71.2 pounds) at 48 weeks, a result that, if replicated in registrational trials, would redefine the efficacy ceiling for the class. An NDA filing is expected in late 2026 or early 2027, with FDA approval potentially in late 2027 or the first half of 2028.
The glucagon receptor component is what distinguishes retatrutide from everything else in development. Glucagon activation increases energy expenditure and fat oxidation, adding a calorie-burning mechanism on top of the appetite suppression delivered by GLP-1 and GIP agonism. If the safety profile holds through Phase 3 completion, retatrutide would give Lilly a generational lead: a next-generation drug arriving before most competitors have even received first approvals for their current-generation candidates.
Medicare Opens the Door: July 1, 2026
The most significant near-term catalyst for the entire GLP-1 market is the Medicare GLP-1 Bridge Program launching July 1, 2026. For the first time, Medicare Part D will broadly cover anti-obesity medications, with beneficiary copays set at $50 per month. The program covers Wegovy (pill and injectable), Zepbound, and Foundayo. Eligibility requires either a BMI of 27 or above with a comorbidity such as heart disease or prediabetes, or a BMI of 35 or above with no additional criteria. The program runs through December 31, 2027.
On the Medicaid side, CMS launched the BALANCE model in December 2025, a voluntary five-year program for state agencies. As of January 2026, only 13 state Medicaid programs cover GLP-1s for obesity under fee-for-service. The enrollment window for states runs from May 2026 through January 2027. Coverage expansion at both the federal and state level represents a structural increase in the addressable patient population.
The Access Problem: Coverage Is Not Universal
Despite the Medicare expansion, access remains uneven. Only 43% of large employers (5,000 or more employees) cover GLP-1s for weight loss. Commercial coverage for Wegovy has actually deteriorated: 42% more patients lacked coverage in early 2026 compared to 2025, with over 41 million Americans without access. Zepbound saw a 12% increase in uncovered patients, exceeding 109 million. Over 88% of patients with coverage still face prior authorization requirements.
The oral formulations may shift this dynamic. Foundayo at $149 per month self-pay and Wegovy pill at comparable pricing eliminate the injection barrier and reduce pharmacy handling costs. If oral GLP-1s demonstrate similar long-term efficacy to injectables, they could expand the total addressable market by reaching patients who would never have accepted a weekly injection.
Market Size: $82 Billion Today, Somewhere Between $95 Billion and $268 Billion by 2030
The range of market size estimates tells its own story. The total GLP-1 market including diabetes and obesity was approximately $82 billion in 2026. Goldman Sachs projects $95 billion by 2030, factoring in pricing pressure, discontinuation rates, and access barriers. Morgan Stanley estimates $190 billion by 2035. BCC Research projects $268 billion by 2030. The spread reflects fundamental uncertainty about how fast the market will expand beyond early adopters and into the broader population where access, affordability, and long-term adherence remain open questions.
What the Numbers Reveal
The competitive picture heading into the second half of 2026 is defined by three dynamics. First, Lilly is pulling away. A 60-40 market share split, a $2 billion guidance raise, the only approved oral GLP-1, and a triple agonist pipeline candidate with nearly 29% weight loss in Phase 3 give Lilly structural advantages that compound over time. Second, Novo is pivoting to oral. The Wegovy pill's $354 million debut, doubling estimates, shows the oral form factor can scale. But CagriSema's head-to-head loss against tirzepatide removes the pipeline narrative that was supposed to close the gap. Third, the challengers are real but distant. Amgen, Roche, Viking, and Structure all have differentiated programs, but none will reach market before 2028 at the earliest. By then, Lilly may have retatrutide approved and a two-generation lead.
The GLP-1 market is not winner-take-all. Dosing convenience, oral versus injectable formulation, side effect profiles, and insurance coverage will create room for multiple products. But the financial and clinical data from Q1 2026 make clear that this is Lilly's race to lose.
Key Figures at a Glance
- Eli Lilly Q1 2026: total revenue $19.8B (+56% YoY), adjusted EPS $8.55 (+156%), full-year guidance raised to $82-85B
- Mounjaro + Zepbound combined Q1 revenue: $12.8B, with Mounjaro at $8.66B and Zepbound at $4.16B
- Foundayo (orforglipron): FDA approved April 1, 2026; 20,000+ patients treated in first weeks; $149/month self-pay
- Lilly US obesity + diabetes market share: 60.1%
- Novo Nordisk Q1 2026: total revenue $10.8B, adjusted EPS $1.03; full-year guide: adjusted sales decline 4-12% at CER
- Wegovy pill: $354M in first quarter, doubled analyst estimates; 1.3M prescriptions in Q1
- CagriSema vs tirzepatide (REDEFINE 4): 23.0% vs 25.5% weight loss — failed non-inferiority
- Retatrutide (Lilly triple agonist): 28.7% weight loss in TRIUMPH-4 Phase 3; NDA filing expected late 2026 / early 2027
- Medicare GLP-1 Bridge Program: launches July 1, 2026; $50/month copay for Part D beneficiaries
- Total GLP-1 market 2026: approximately $82B; 2030 estimates range from $95B (Goldman Sachs) to $268B (BCC Research)
For a detailed breakdown of Eli Lilly's Q1 2026 earnings, including segment-level performance and management commentary on the GLP-1 franchise trajectory, the full episode is available at betafinch.com/podcasts/lly.